Hottest science and technology news from the United States
Provided by AGPCorvus Pharmaceuticals Presents Soquelitinib Phase 1 Atopic Dermatitis Data at the Society for Investigative Dermatology (SID) Annual Meeting
New immunologic and biomarker data supports the potential of ITK inhibition with soquelitinib to increase persistent Treg cells and influence multiple inflammatory pathways.
Data supports potential for soquelitinib to rebalance the immune system and produce drug-free remissions following discontinuation of therapy
Immunologic markers consistent with soquelitinib’s novel mechanism of action and clinical results
SID data included in late-breaking oral presentation; company to review data at investor and analyst meeting today at 1:30 pm ET (12:30 pm CT)
SOUTH SAN FRANCISCO, Calif., May 14, 2026 (GLOBE NEWSWIRE) -- Corvus Pharmaceuticals, Inc. (Nasdaq: CRVS), a clinical-stage biopharmaceutical company, announced today the presentation of final data from its randomized, blinded, placebo-controlled Phase 1 trial evaluating soquelitinib in patients with moderate-to-severe atopic dermatitis in two oral sessions at the Society for Investigative Dermatology (SID) Annual Meeting, which is taking place May 13-16, 2026 in Chicago. The first oral presentation covers soquelitinib’s immunologic and clinical activity, and the second late-breaking oral presentation covers its potential to provide prolonged drug-free remissions.
“While there are a number of approved systemic options for atopic dermatitis, they don’t work for all patients and they are limited by the need for prolonged dosing regimens, rapid disease rebound following cessation of therapy, or inconvenient drug administration and monitoring,” said Albert S. Chiou, M.D., MBA, Clinical Professor, Dermatology and Director of Clinical Research in the Department of Dermatology at Stanford University Medical Center. “There is a need for a safe oral systemic therapy that can address these limitations and be effective across the disease spectrum including relapsed/refractory patients. The results of this trial are exciting as they suggest that soquelitinib may meet this need, with the possibility of offering shorter treatment regimens and prolonged disease clearance for patients.”
The trial enrolled 72 patients with moderate-to-severe atopic dermatitis that previously failed one prior topical or systemic therapy. Thirty-five percent of the patients had received a prior systemic therapy. In each of cohorts 1-3, 16 patients were randomized in a 3:1 ratio to receive one of three 28-day dosing regimens (100 mg twice daily, 200 mg once daily or 200 mg twice daily) of soquelitinib or equivalent placebo. In cohort 4, 24 patients were randomized in a 1:1 ratio to receive a 56-day, 200 mg twice daily dosing regimen of soquelitinib or equivalent placebo. Patients in the study were monitored for a 30-day post treatment follow up period, except in cohort 3, which had an extended 90-day post treatment follow up period.
Clinical Highlights from Soquelitinib Phase 1 Atopic Dermatitis Clinical Trial
The data demonstrated safety and positive efficacy results, including in patients who received prior systemic therapy and were treatment resistant. In addition, there was a dose dependent efficacy trend in cohorts 1-3, and additional clinical benefit was observed with longer treatment in cohort 4. Across all four cohorts, no significant safety issues were observed. Reported adverse events occurred in 41.7% of soquelitinib patients and 41.7% of placebo patients; all were Grade 1-2 and did not result in any dose modifications or interruptions. No severe or serious adverse events were reported. No significant lab abnormalities were seen.
Key efficacy results from cohorts 3 and 4 (200 mg twice daily) include:
- Cohort 3: 50% of soquelitinib patients achieved EASI 75, 8% achieved EASI 90 and 25% achieved IGA 0/1
- Cohort 4: 75% of soquelitinib patients achieved EASI 75, 25% achieved EASI 90 and 33% achieved IGA 0/1
- In both cohorts 3 and 4, disease control continued throughout the post treatment follow up periods (90 and 30 days, respectively), see Figure 1 below; this compares to published data from approved systemic therapies that show a worsening or rebound of disease usually occurring within four weeks of stopping therapy and in some instances requiring the need for rescue medication.
Figure 1 below shows the percent reduction in EASI for cohort 3 (left) and cohort 4 (right). No disease rebound was observed and no rescue medications were required.
Figure 1: Soquelitinib Treatment Leads to Durable Remissions with No Rebound Following Four and Eight Weeks of Treatment.

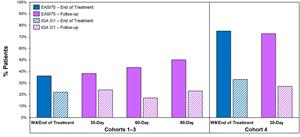
Figure 2 below shows the durability of remission in patients achieving EASI 75 and IGA 0/1 in cohorts 3 and 4. The percent of patients achieving EASI 75 and IGA0/1 is maintained during the drug-free follow up period.
Figure 2: Percentage Patients Achieving EASI 75 and IGA 0/1.
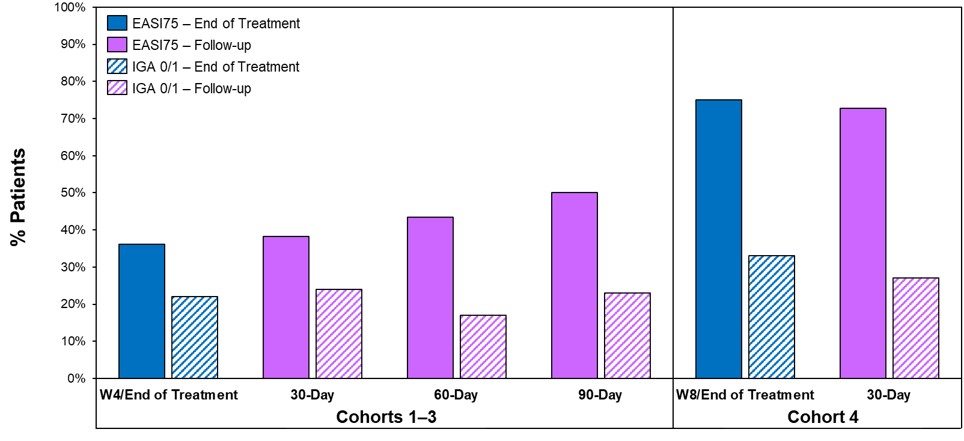
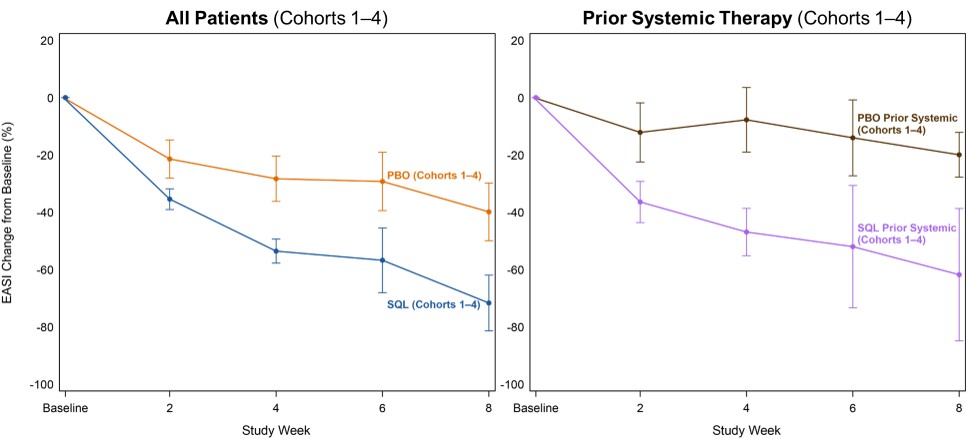
Figure 3 below shows the percent reduction in EASI scores for all patients (left) in cohorts 1-4 and the EASI curves for patients who had received prior systemic therapies (right). The data shows comparable efficacy in patients with prior systemic therapy and the all patients group.
Figure 3: Efficacy in Patients with Prior Systemic Therapy (Cohorts 1–4).
Biomarker Highlights and Immunologic Studies from Soquelitinib Phase 1 Atopic Dermatitis Clinical Trial
Soquelitinib is designed to specifically bind and inhibit ITK in T cells, modulating T cell signaling and differentiation. This results in the blockade of Th2 and Th17 cell function and their secretion of various inflammatory cytokines such as IL-4, IL-5, IL-13, IL-17 and others. ITK also regulates a switch from Th17 cell differentiation to T regulatory (Treg) cells, which suppress inflammation and autoimmunity. Inhibition of ITK by soquelitinib produces a reduction of Th17 and an increase in Treg function. Immunologic and molecular studies on peripheral blood taken from patients in the study were conducted and support soquelitinib’s mechanism of action, including the potential for drug-free remissions:
- Blocking Th2 and Th17 function and their related downstream cytokines (IL-4, IL-5, IL-13). See Figures 4a and 4b below.
- Increasing persistent Treg cells, both during treatment and in the post-treatment, drug-free period, by regulating the switch to Treg cells from Th17 cells. See Figures 5a and 5b below.
- Inhibiting JAK-STAT signaling, upregulating SOCS3 (suppressor of cytokine signaling 3 protein, a negative regulator of JAK-STAT signaling) and reducing JAK1 and STAT6. See Figures 6a and 6b below.
Together, these immunomodulatory effects reduce inflammation and restore immune balance by enhancing Treg function. The observed clinical manifestations of these effects are 1) prompt responses; 2) clinical responses that are durable without rebound; and 3) specificity of soquelitinib for ITK confers safety without immunosuppression.
“The biomarker data demonstrates the translation between soquelitinib’s mechanism of action and its clinical activity. The immunologic markers are consistent with soquelitinib’s mechanism of action and the clinical results from the Phase 1 trial,” said Kavita Sarin, M.D., Ph.D., Professor of Dermatology and Director of the Stanford Skin Cancer Genetics Program at the Stanford Cancer Institute. “The data supports the effects of soquelitinib on multiple T cell properties including cytokine production, inflammatory cell function and the induction of persistent Tregs. The data also provides support for the findings of the lack of disease rebound observed in treated patients.”
Figure 4a below shows a dose dependent reduction in proliferating circulating Th2 cells measured by scRNAseq in blood from patients evaluated in cohorts 1-3 receiving placebo, soquelitinib 200 mg once a day or 200 mg twice per day. Each dot represents a patient with changes compared to baseline. (Ki-67, proliferation marker; GATA3, c-MAF, CCR4 and PTGDR2 are Th2 markers; STAT6 is involved in inflammatory signaling.)
Figure 4a: Dose-dependent Reduction in Proliferating Th2*.
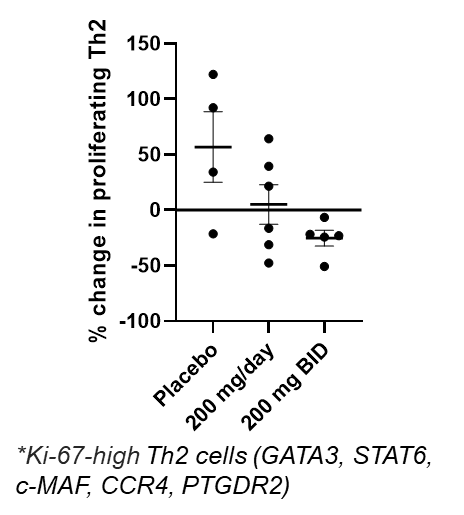
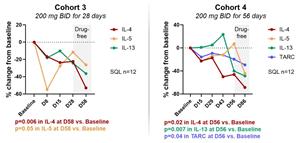
Figure 4b below shows median values of serum cytokines measured over time from cohorts 3 and 4. The decrease in levels of Th2 cytokines and TARC continue after the treatment is discontinued (drug-free period).
Figure 4b: Reduction in IL-4, IL-5, IL-13 and TARC.
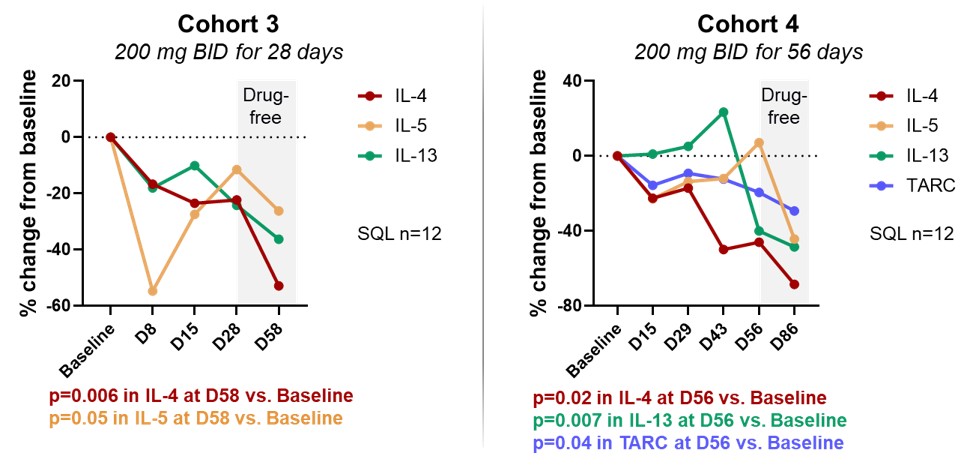
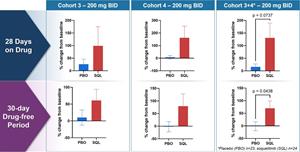
Figure 5a below shows percent change from baseline in circulating Treg cells for placebo (N=23 for cohort 1-4 combined) and treated patients (N=24) from cohort 3, cohort 4 and combined. The top row shows values after 28 days of therapy and the bottom row shows values 30 days after stopping therapy (drug-free period).
Figure 5a: Soquelitinib Treatment Leads to Increase in Persistent Tregs (CD4+, Foxp3+, CD25Hi ) measured by flow cytometry.
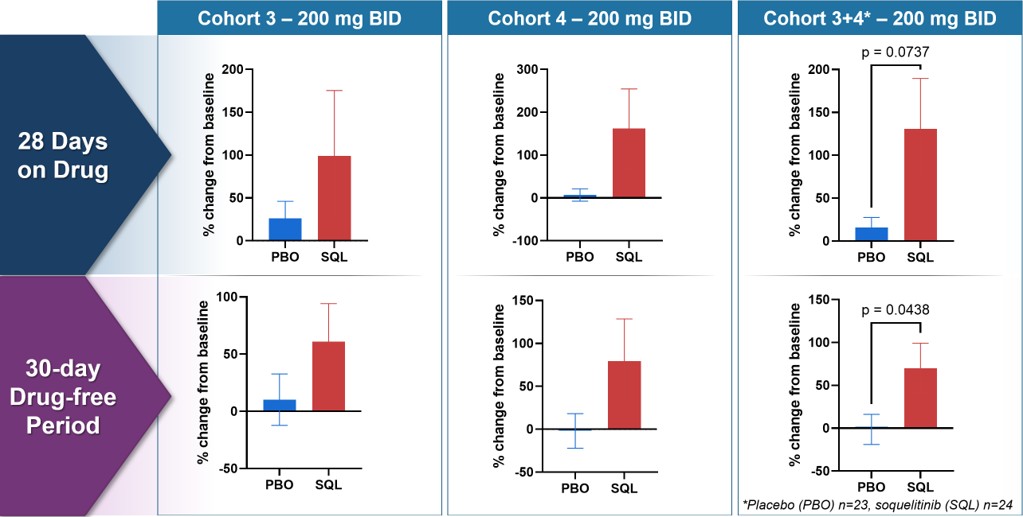
Figure 5b below shows changes from baseline for markers of Tregs and Th17 cells (patients as in Figure 4). BACH2 is a transcription factor that promotes Treg function and stability by binding to Foxp3 promoter. RORγT is the master transcription factor for Th17 cell function. The data demonstrates an increase in Tregs and a decrease in Th17, consistent with soquelitinib induced “switch.”
Figure 5b: Inhibiting ITK Regulates Switch to Treg from Th17 - Increased BACH2 and Reduced RORγT Expression measured using scRNAseq.
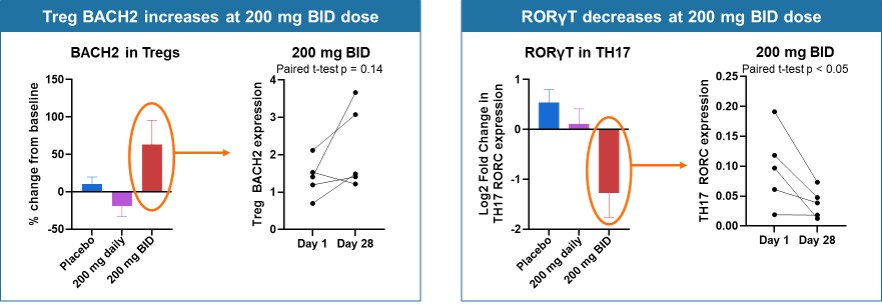
Figure 6a below shows percent change from baseline of SOCS3 (suppressor of cytokine signaling-3) in Th2, Th1 and Tc17 cells for placebo and treated patients from cohorts 1-3. SOCS3 regulates cytokine signaling in the JAK/STAT pathway, with increased levels of SOCS3 inhibiting T cell responses to cytokines. Tc17 cells produce IL-17, demonstrate high plasticity, are driven by transcription factor RORγT and are important in autoimmune diseases. These findings indicate that soquelitinib will reduce IL-17 production.
Figure 6a: SOCS3 Increases in Circulating T cells.
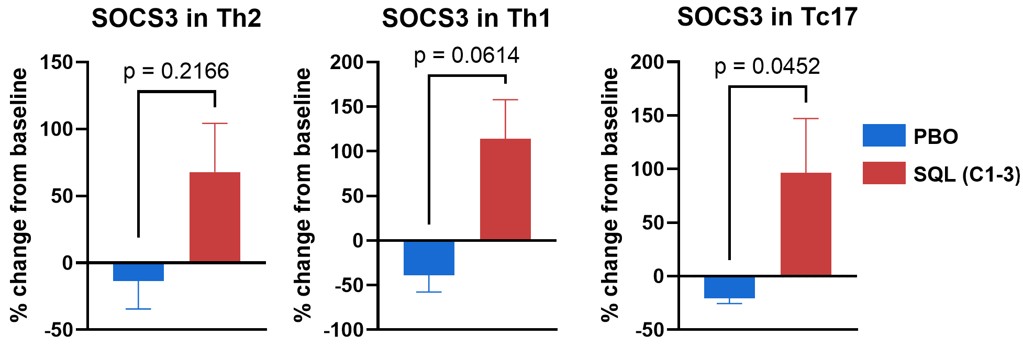
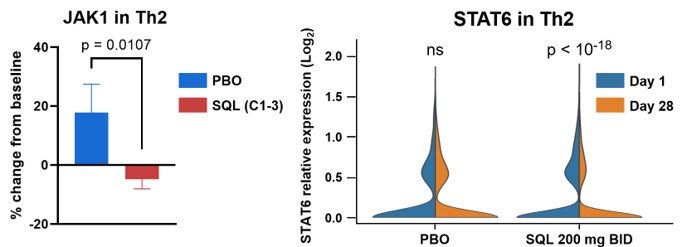
Figure 6b below shows change from baseline (Cohorts 1-3) in JAK1 and STAT6 resulting from treatment with soquelitinib compared to placebo. The violin plots on the right show a reduction in STAT6 expression at day 28 (gold) compared to baseline (blue) for the 200 mg twice daily (N=5) patients; no significant change was seen in placebo (N=4) patients.
Figure 6b: JAK1 and STAT6 Decreases in Circulating Th2 Cells measured by scRNAseq.
“The soquelitinib Phase 1 data shows the potential of ITK inhibition to treat a broad range of atopic dermatitis patients, including those who are resistant to other systemic therapies,” said Richard A. Miller, M.D., co-founder, president and chief executive officer of Corvus. “Biomarker data supports soquelitinib’s novel mechanism of action, demonstrating the ability to modulate several key cellular functions that are not currently targeted by approved and in development systemic therapies. The data also supports the potential for soquelitinib to be a novel treatment for a range of autoimmune and inflammatory diseases based on a rebalancing of immunity, which could reduce the need for chronic long-term therapy. We believe that soquelitinib may have the potential to change the underlying biology of these diseases.”
Soquelitinib Phase 2 Atopic Dermatitis Clinical Trial
Corvus is currently enrolling patients in the soquelitinib SIERRA1 Phase 2 randomized placebo-controlled clinical trial that is anticipated to enroll approximately 200 patients with moderate-to-severe atopic dermatitis that have failed at least one prior topical or systemic therapy. This includes four cohorts of 50 patients each, with soquelitinib doses of 200 mg once per day, 200 mg twice per day and 400 mg once per day, along with a placebo group. The treatment period is 12 weeks with a 90-day follow-up period with no treatment.
Angel Pharmaceuticals, Corvus’ partner in China, also is enrolling a Phase 1b/2 clinical trial evaluating soquelitinib in patients with moderate-to-severe atopic dermatitis. This is a blinded, placebo-controlled trial that is planned to evaluate a 12-week treatment regimen in 48 patients utilizing soquelitinib doses of 100 mg twice per day, 200 mg once per day, 200 mg twice per day and 400 mg once per day. The patient eligibility and endpoints are similar to those used previously by Corvus. Depending on the results from the Phase 1b portion of the study, an additional 60-90 patients will be enrolled in the Phase 2 portion of the study. The trial is open at several leading dermatology centers in China who have been involved in global registration trials. The study is conducted in close collaboration with Corvus. Results from the initial cohorts are anticipated late this year.
Society for Investigative Dermatology Annual Meeting Presentations
The SID oral presentation slides are available on the Publications and Presentations page of the Corvus website. The presentation details are as follows:
Oral Presentation
Title: Immunologic and clinical activity of soquelitinib, a selective ITK inhibitor, in atopic dermatitis
Abstract #: 0483
Date / Time: May 14, 2026, 8:45 - 8:55 am CT
Presenter: Kavita Sarin, M.D., Ph.D.
Late-Breaking Oral Presentation
Title: Soquelitinib, an ITK inhibitor, Produces Prolonged Drug-Free Remissions in Atopic Dermatitis
Abstract #: LB1154
Date / Time: May 16, 2026, 10:40 - 10:50 am CT
Presenter: Albert Chiou, M.D.
Investor and Analyst Meeting
Corvus will review the soquelitinib data presented at SID during an investor and analyst meeting taking place today, Thursday, May 14, 2026 from 1:30 – 2:30 pm ET (12:30 – 1:30 pm CT). The meeting will be hosted by Dr. Miller and will feature Dr. Chiou and Dr. Sarin as guest speakers. The meeting will be available as a live webcast (including presentation slides) via the investor relations section of the Corvus website. A replay of the webcast will be available on Corvus’ website.
About Corvus Pharmaceuticals
Corvus Pharmaceuticals is a clinical-stage biopharmaceutical company pioneering the development of ITK inhibition as a new approach to immunotherapy for a broad range of immune diseases and cancer. The Company’s lead product candidate is soquelitinib, an investigational, oral, small molecule drug that selectively inhibits ITK. Soquelitinib is being evaluated in a registration Phase 3 clinical trial for relapsed/refractory PTCL and in a Phase 2 clinical trial for the treatment of atopic dermatitis. Its other clinical-stage candidates are being developed for a variety of cancer indications. For more information, visit www.corvuspharma.com or follow the Company on LinkedIn.
About Soquelitinib
Soquelitinib (formerly CPI-818) is an investigational small molecule drug given orally designed to selectively inhibit ITK (interleukin-2-inducible T cell kinase), an enzyme that is expressed predominantly in T cells and plays a role in T cell and natural killer (NK) cell immune function. Soquelitinib has been shown to affect T cell differentiation and induce the generation of Th1 helper cells while blocking the development of both Th2 and Th17 cells and production of their secreted cytokines. Th1 T cells are required for immunity to tumors, viral infections and other infectious diseases. Th2 and Th17 helper T cells are involved in the pathogenesis of many autoimmune and allergic diseases. Recent studies have demonstrated that ITK controls a switch between the differentiation of Th17 proinflammatory cells and T regulatory suppressor cells. Inhibition of ITK leads to a shift toward T regulatory cell differentiation, which has the potential to suppress autoimmune and inflammatory reactions. The Company believes the inhibition of specific molecular targets in T cells may be of therapeutic benefit for patients with autoimmune and allergic diseases and in cancers, including solid tumors. Based on interim results from a Phase 1/1b clinical trial in patients with refractory T cell lymphomas, which demonstrated tumor responses in very advanced, refractory, difficult to treat T cell malignancies, the Company is enrolling a registration Phase 3 clinical trial (NCT06561048) of soquelitinib in patients with relapsed/refractory PTCL. Soquelitinib is also now being investigated in a randomized placebo-controlled Phase 2 clinical trial in patients with atopic dermatitis. A publication describing the chemistry, enzymology and biology of soquelitinib appeared in npj Drug Discovery in December 2024 and is available online at the Nature website and on the Publications and Presentations page of the Corvus website.
About Atopic Dermatitis
Atopic dermatitis, also called eczema, is a chronic disease that can cause inflammation, redness, scaly patches, blisters and irritation of the skin. It affects up to 20% of children and up to 10% of adults, and treatments include topical therapies, oral therapies and systemic injectable biologic therapies. It is frequently associated with other allergic disorders such as food allergies and asthma. Atopic dermatitis, like asthma and allergy, involves the participation of Th2 lymphocytes which secrete cytokines that result in inflammation. Soquelitinib has been shown in preclinical and clinical studies to inhibit cytokine production from Th2 lymphocytes.
About Angel Pharmaceuticals
Angel Pharmaceuticals is a privately held biopharmaceutical company developing a pipeline of precisely targeted investigational medicines for cancer, autoimmune, infectious and other serious diseases in China. Angel Pharmaceuticals was launched through a collaboration with Corvus and investments from investors in China. Angel Pharmaceuticals licensed the rights to develop and commercialize Corvus’ three clinical-stage candidates – soquelitinib, ciforadenant and mupadolimab – in greater China and obtained global rights to Corvus’ BTK inhibitor preclinical programs. Under the collaboration, Corvus currently has a 49.7% equity stake in Angel Pharmaceuticals excluding 7% of Angel’s equity reserved for issuance under the Angel employee stock ownership plan, and Corvus has designated three individuals on Angel’s five-person Board of Directors. For more information, visit www.angelpharma.com.
Forward-Looking Statements
This press release contains forward-looking statements, including statements related to soquelitinib’s potential to provide prolonged drug-free remissions and shorter treatment regimens; the potential safety and efficacy of the Company’s product candidates; the potential use of soquelitinib to treat a range of autoimmune and inflammatory diseases and to change the underlying biology of these diseases; the Company’s leadership position; and clinical strategy and the design of clinical trials, including the Company’s collaborations and the timeline for initiation, target or expected number of patients to be enrolled, dose levels, number of sites and other product development milestones. All statements other than statements of historical fact contained in this press release are forward-looking statements. These statements often include words such as “believe,” “expect,” “anticipate,” “intend,” “plan,” “estimate,” “seek,” “will,” “may” or similar expressions. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. The Company’s actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to, risks detailed in the Company’s Quarterly Report on Form 10-Q for the quarter ended March 31, 2026, filed with the Securities and Exchange Commission on May 7, 2026, as well as other documents that may be filed by the Company from time to time with the Securities and Exchange Commission. In particular, the following factors, among others, could cause results to differ materially from those expressed or implied by such forward-looking statements: the Company’s ability to demonstrate sufficient evidence of efficacy and safety in its clinical trials of its product candidates; the accuracy of the Company’s estimates relating to its ability to initiate and/or complete preclinical studies and clinical trials and release data from such studies and clinical trials; the results of preclinical studies and interim data from clinical trials not being predictive of future results; the Company’s ability to enroll sufficient numbers of patients in its clinical trials; the unpredictability of the regulatory process; regulatory developments in the United States and foreign countries; the costs of clinical trials may exceed expectations; the Company’s ability to accurately estimate the cash on hand providing funding into the second quarter of 2028 and the Company’s ability to raise additional capital. Although the Company believes that the expectations reflected in the forward-looking statements are reasonable, it cannot guarantee that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and the timing of events and circumstances and actual results could differ materially from those projected in the forward-looking statements. Accordingly, you should not place undue reliance on these forward-looking statements. All such statements speak only as of the date made, and the Company undertakes no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise.
INVESTOR CONTACT:
Leiv Lea
Chief Financial Officer
Corvus Pharmaceuticals, Inc.
+1-650-900-4522
llea@corvuspharma.com
MEDIA CONTACT:
Julia Stern
Real Chemistry
+1-763-350-5223
jstern@realchemistry.com
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/1dfb622c-604b-4e02-9d90-d36c6719bfa1
https://www.globenewswire.com/NewsRoom/AttachmentNg/424e5145-89b3-4b7d-b292-55a439070856
https://www.globenewswire.com/NewsRoom/AttachmentNg/aacc680b-c139-4cf8-a14f-4a13e2a07f5b
https://www.globenewswire.com/NewsRoom/AttachmentNg/4141ea89-721a-4e99-b8f2-7904e8ff569d
https://www.globenewswire.com/NewsRoom/AttachmentNg/0233bb9f-6e54-4df1-9f91-5aa7bd93517c
https://www.globenewswire.com/NewsRoom/AttachmentNg/e2bed082-4a20-4a9b-820b-c06b2a251b71
https://www.globenewswire.com/NewsRoom/AttachmentNg/31a9a903-a2f9-433f-8789-59aca2d6e4c2
https://www.globenewswire.com/NewsRoom/AttachmentNg/2725858e-6afa-4f03-9b80-16ebb061c711
https://www.globenewswire.com/NewsRoom/AttachmentNg/15ebe481-1b5c-46d2-a16f-e3324d323184

Figure 1
Soquelitinib Treatment Leads to Durable Remissions with No Rebound Following Four and Eight Weeks of Treatment.
Figure 2
Percentage Patients Achieving EASI 75 and IGA 0/1.
Figure 3
Efficacy in Patients with Prior Systemic Therapy (Cohorts 1–4).
Figure 4a
Dose-dependent Reduction in Proliferating Th2*.
Figure 4b
Reduction in IL-4, IL-5, IL-13 and TARC.
Figure 5a
Soquelitinib Treatment Leads to Increase in Persistent Tregs (CD4+, Foxp3+, CD25Hi ) measured by flow cytometry.
Figure 5b
Inhibiting ITK Regulates Switch to Treg from Th17 - Increased BACH2 and Reduced RORγT Expression measured using scRNAseq.
Figure 6a
SOCS3 Increases in Circulating T cells.
Figure 6b
JAK1 and STAT6 Decreases in Circulating Th2 Cells measured by scRNAseq.
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.